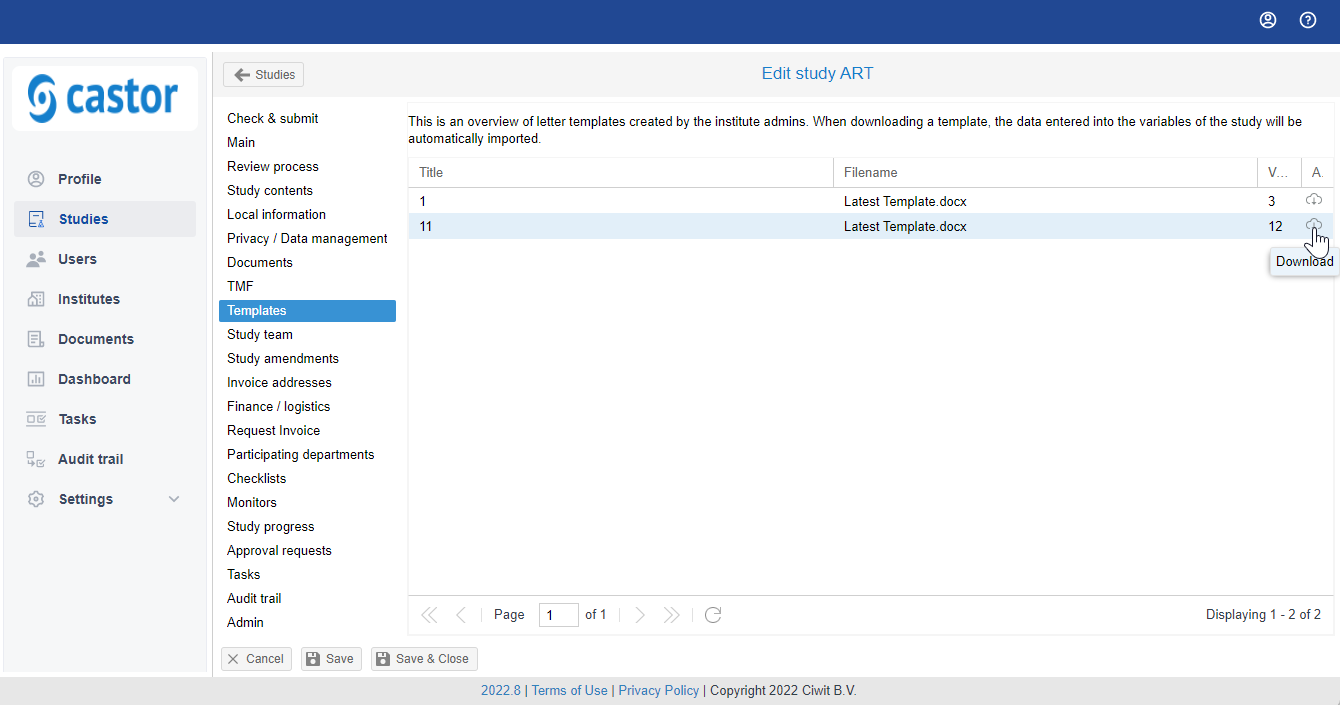
Automatically generate a letter in SMS
Admin users can add templates to Castor SMS for letters, which include study variables. When downloading such a letter template from a study, the entered values from the system will automatically be imported into the letter.
Steps:
- Open the study and navigate to the "Templates" tab
- Click on the download icon or right click with your mouse to download the letter