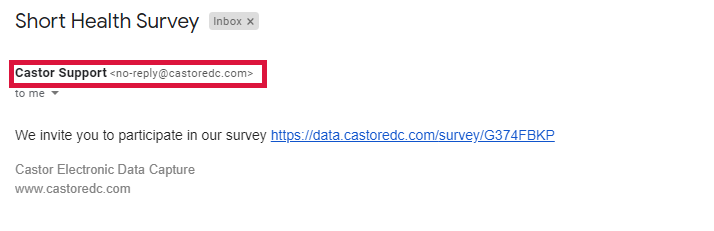
Why are survey invitations sent from no-reply@castoredc.com in EDC/CDMS?
The standard address from which surveys are sent is no-reply@castoredc.com. This will always appear as the sender email address.
When you select a user email address as the sender, the participant (receiver) will still receive an email from no-reply@castoredc.com, but the name of the user will appear as sender.
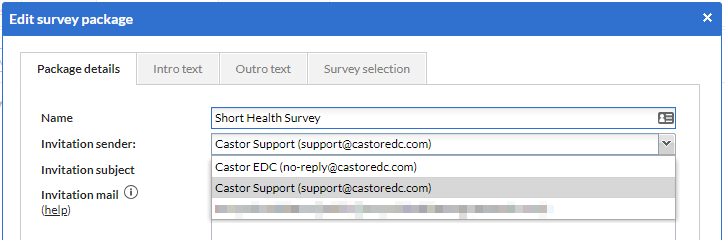
In addition, the survey participant can reply to the email and the reply will be sent to the selected sender email address.